|
5/29/2023 0 Comments Chromium iii sulfide formula 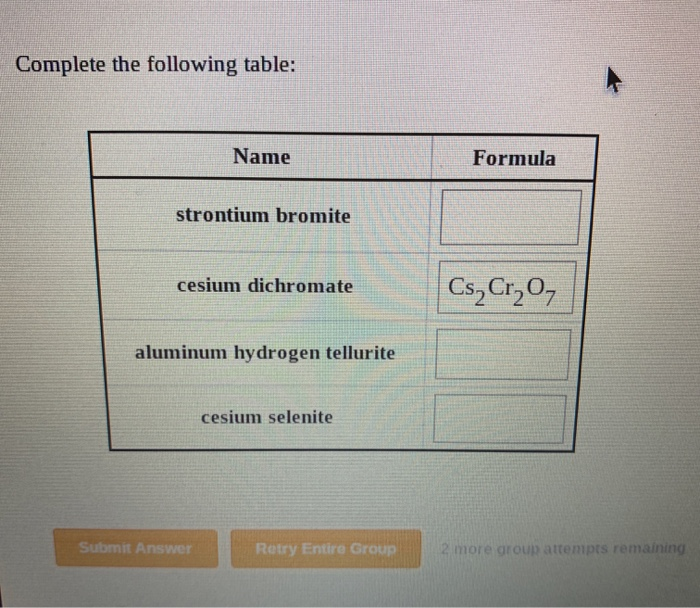
5 Interesting Facts About Green Chrome Oxide – Basstechintl.Stoichiometry of Ammonium Dichromate Decomposition –.It can also cause reproductive toxicity and is harmful to the unborn child. Sulfur needs two electrons to be like Argon, the next noble gas. 401 CoS cobalt (ii) sulfide 25.00 5.4500 1 322 CdMoO4 cadmium molybdate 1. You can figure this out from the Periodic Table. 134885-25G Catalog Description, Chromium(III) sulfide, 99 trace metals basis MDL Number, MFCD00010950 Master Molecular formula, Cr2S3 Molecular formula. It may cause an allergic skin reaction and serious eye damage. Density of Solid Density of Solid ID FORMULA NAME CAS No T. Įxposure to chromium (iii) oxide through mouth and skin can cause acute toxicity. It serves as an abrasive for polishing or stropping the edges of razors, knives, and surfaces of optical devices.Because of its stability, chromium (iii) oxide is used as the green pigment in inks, glasses, and paints and as a colorant for ceramics, as well as producing a faint green tinge in glazes.Practically insoluble, 2.96 µg/L (20 ☌, pH 8), 3.13 µg/L (20 ☌, pH 6) The three waters added back in balance the change from ammonia to ammonium as well as the three hydroxides on the chromium(III) hydroxide. Insoluble in acetone and alcohol, very slightly soluble in alkalies and acids When chromium (iii) oxide reacts with gaseous hydrogen sulfide, it yields chromium (iii) sulfide and water:Ĭr 2O 3 + 3H 2S → Cr 2S 3 + 3H 2O Properties and Characteristics of Chromium (III) Oxide General Propertiesĭry, light or dark green, fine crystalline powder It reacts with dilute hydrochloric acid to form chromium (iii) chloride and water :Ĭr 2O 3 + 6HCl → 2CrCl 3 + 3H 2O Chromium (III) Oxide with Hydrogen Sulfide Ammonium dichromate, on heating, decomposes into chromium (iii) oxide, water, and nitrogen :Ĭhromium (iii) carbonate is heated to form chromium (iii) oxide and carbon dioxide:Ĭr 2(CO 3) 3 → Cr 2O 3 + 3CO 2 Reaction with Other Compounds Chromium (III) Oxide with Hydrochloric Acid 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
